|
Take heed to this text |

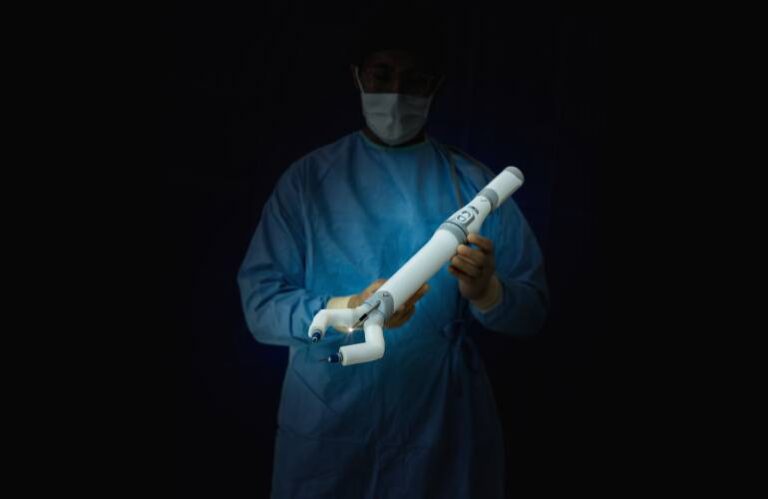
MIRA includes a small, self-contained surgical system that may carry out stomach surgical procedures. | Supply: Digital Incision
Digital Incision introduced that the FDA granted advertising and marketing authorization to its MIRA miniaturized surgical robotic system.
The FDA gave the nod for using the miniRAS (robotic-assisted surgical procedure) system in adults present process colectomy procedures. MIRA went by the FDA’s de novo classification course of, with findings primarily based on an investigational system exemption (IDE) examine.
Digital Incision’s MIRA system includes a small, self-contained surgical system. Inserted by a single midline umbilical incision within the affected person’s stomach, it permits for complicated, multi-quadrant stomach surgical procedures. The system additionally makes use of present minimally invasive instruments and methods which might be acquainted to surgeons. Altogether, MIRA weighs about two kilos.
Based on the corporate, the system’s tray-to-table design may present the benefits of robotic surgical procedure with out requiring them to arrange the working room across the system — a present problem with surgical robots.
“At present marks a turning level in surgical robotics as we have now hit a major milestone in making miniaturized robotic surgical procedure a actuality,” mentioned Digital Incision President and CEO John Murphy. “For greater than a decade, our group has been devoted to our core mission of creating each working room robot-ready. I’m extremely proud to see our efforts come to fruition. We lengthen our gratitude to the FDA for its considerate overview of MIRA’s know-how and our medical proof.
“Whether or not as a complement to the present mainframes or as a stand-alone platform, miniaturization has the potential to speed up the adoption of robotic-assisted surgical procedure.”
Hear from Digital Incision through the Surgical Robotics Week sequence subsequent month.
How the Digital Incision MIRA system acquired right here
The MIRA platform acquired IDE approval in October 2020, adopted by approval for an IDE complement in April. Digital Incision submitted MIRA to the FDA for de novo overview in Could 2023. Notably, MIRA went to house final month, with NASA awarding a grant to make use of the system on a 2024 know-how demonstration mission.
In November 2021, Digital Incision accomplished a $46 million Sequence C financing spherical to assist the robotic surgical procedure platform. Digital Incision then raised $30 million extra in September 2023.
Digital Incision plans to start commercialization of MIRA by a primary entry program throughout choose U.S. facilities. It plans to ramp up manufacturing and increase to extra websites over time. The corporate additionally hopes to pursue extra indications, together with gynecology, common surgical procedure, urology and different comfortable tissue and strong organ surgical procedure.
It plans for gynecological procedures in 2024. Moreover, the corporate has a brand new iteration of the know-how tailor-made for common surgical procedure in its design stage. It expects first-in-human makes use of outdoors the U.S. later this 12 months.
Editor’s Be aware: This text was syndicated from The Robotic Report’s sister website MassDevice.